LABORATORY TESTING SERVICES
Lab 797 works in conjunction with individual compounding facilities as well as certification companies across the country to perform environmental testing in cleanrooms and other designated areas. Specializing in microbial identification, Lab 797 offers reliable, cost-effective testing with quick turn-around times. Lab 797 is an ISO/IEC 17025:2017 accredited laboratory through the ANSI National Accreditation Board. Let us help you create custom solutions for your pharmacy, cleanroom, or other facility! See our service offerings below.
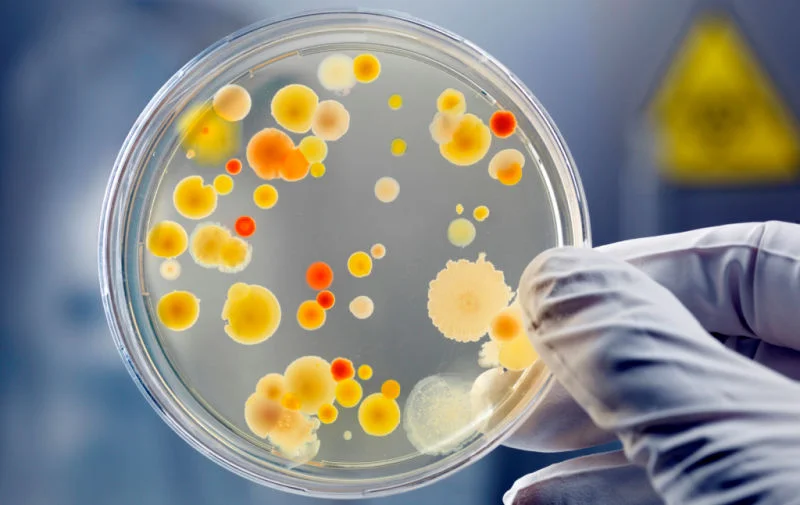
USP 797 and Microbiology
Services
USP 797 is a chapter of the United States Pharmacopeia that sets the standards for sterile compounding in healthcare and pharmacies. Requirements include cleanroom and environmental control, personnel training, and environmental monitoring. We can assist with all requirements to make sure your facility is compliant with USP 797.
Not a pharmacy? We offer microbiology services for any industry. Please contact us for more information on custom microbiology solutions.
Organism Identification
Identification services are available for most bacterial and fungal organisms recovered from environmental samples. This ensures that no highly pathogenic organisms are present in your classified areas and can assist with creating meaningful action plans when samples have out-of-compliance results. Media incubated within your own facility can also be sent to us for identification services.
Incubation and Enumeration
All samples are incubated according to current USP 797 guidelines. Appropriate media and incubation protocols are available for the recovery of both bacteria and fungi. Incubation times may vary based on media type and protocol. Most reports are available within 48-72 hours after incubation, using our secure file sharing software, Sharefile. You can access your reports 24/7 from your facility.
Monthly Sampling Subscription
All the supplies and services required to perform surface and/or personnel sampling monthly in one neat package. Surface and personnel sampling kits are available and ready to ship. Viable airborne monitoring solutions and certifications are available by request. Kits include shipping to and from your facility, sampling instructions, sample processing, organism identification, data trending and free USP 797 consultation. Custom solutions tailored for your pharmacy. Inquire by contacting us by phone or email. Click here to Inquire Today!
Sampling Media Shipped to You
We can deliver sampling materials to your facility fast- with overnight and 2nd day shipping options available. We ship all sampling supplies the same day if placed by 12 PM MST. Tryptic Soy Agar and Sabouraud Dextrose Agar media is available in house. 5 Technician Media Fill Kits are in stock. Other sampling a-la-carte solutions are available by request. Order media here.

Water Testing Services
ANSI/AAMI ST108 Validation and Routine Panels
Validation and routine testing kits come with everything you need to test your critical, utility, and/or steam water. Kits include test vials, cold packaging materials, and return shipping label.
Individual Water Tests
We offer a variety of individual tests for water samples. These include Heterotrophic Plate Count (Bacteria), Endotoxin, Total Hardness, Alkalinity, Conductivity, pH, Total Organic Carbon (TOC) and Ionic tests (Silicate, Copper, Lead, Iron, Manganese, Nitrate, Phosphate, Sulfate, and Zinc).
Dental Clinic Water Testing
Many states require dental clinics to follow CDC guidelines that require regular testing to ensure dental unit water meets the EPA standard of <500 CFU/ml. We provide kits specific for dental clinics to perform this testing.
USP 1231 Water Testing
USP 1231 outlines requirements for the design, validation, operation, and maintenance of pharmaceutical water systems. We can provide testing options to ensure that your water system in compliant with current regulations.
Legionella Testing
Legionella bacteria can grow in building water systems and cause Legionnaire’s disease, a severe form of pneumonia. Routine testing helps detect contamination early, protect patients and staff, and maintain regulatory compliance.
Water is a critical ingredient and utility that directly impacts the quality and safety of products. A variety of industries that use critical or sterile water require laboratory testing. These can include pharmacies, bio-tech, healthcare facilities/hospitals, sterile processors, surgical centers and dental offices. Types of water facilities test include utility water, critical water, or steam.
Depending on your facility, water may need to be tested at different intervals to ensure safety and quality. Not sure where to start? Reach out and inquire with us.
